The Complete Guide to Clinical Project Management: From Concept to Regulatory Success
In today’s highly regulated research environment, successful trials require more than operational execution—they demand strategic oversight, regulatory precision, and cross-functional coordination. Effective clinical project management ensures that every stage of development progresses efficiently while maintaining compliance and data integrity.
At CUREXBIO, integrated lifecycle management supports sponsors from early discovery through regulatory submission and beyond.
Why Clinical Project Management Is Critical in Drug Development
Drug development is a multi-phase process involving scientific validation, regulatory compliance, stakeholder coordination, and financial oversight. Without a structured framework, even promising therapies can face delays, budget overruns, or regulatory setbacks.
Clinical project management provides:
-
Clear scope definition and governance
-
Timeline and milestone tracking
-
Budget allocation and forecasting
-
Risk identification and mitigation
-
Regulatory compliance oversight
-
Cross-functional communication alignment
This structured approach minimizes uncertainty and maximizes operational efficiency across the development lifecycle.
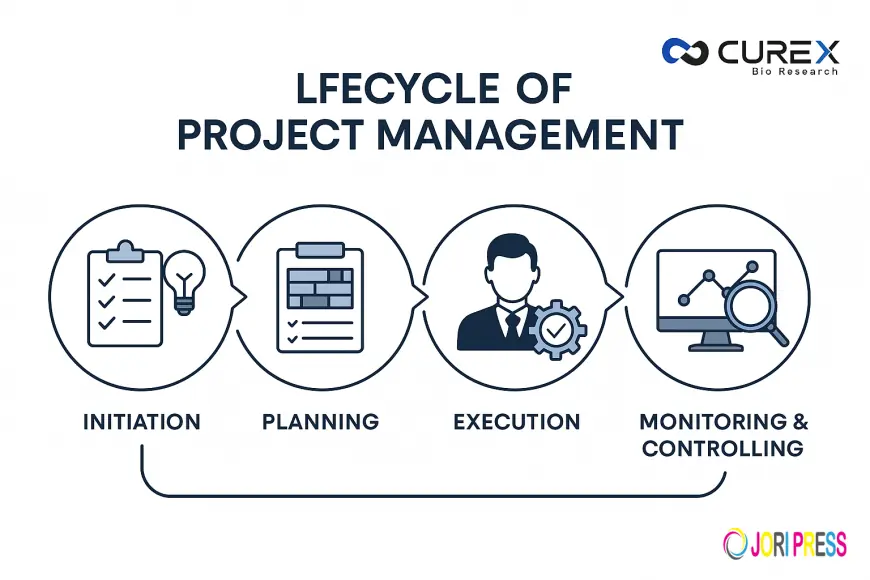
The 5 Phases of Clinical Project Management
1. Initiation: Building the Foundation
The initiation phase defines project objectives, evaluates feasibility, and aligns stakeholders. This includes developing a project charter, conducting gap analyses, and identifying regulatory pathways.
For example, when planning a Phase II oncology trial, teams must align protocols with regulatory standards such as the U.S. Food and Drug Administration and the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use.
Early-stage support often begins with robust preclinical research services, where feasibility, toxicology, and translational planning are established. Learn more about CUREXBIO’s comprehensive Pre-Clinical Support Services.
2. Planning: Designing a Strategic Roadmap
The planning phase converts objectives into actionable strategies. Teams develop:
-
Detailed project timelines (Gantt charts)
-
Budget forecasts
-
Risk management plans
-
Site selection strategies
-
Patient recruitment projections
-
Vendor management frameworks
Strong planning reduces costly delays later in execution. Sponsors benefit from comprehensive clinical development services that streamline study startup, operational logistics, and global trial management.
Explore CUREXBIO’s Clinical Development Services to understand how structured planning improves trial performance.
3. Execution: Bringing the Trial to Life
Execution is where strategy meets action. Activities include:
-
Site activation
-
Investigator training
-
Patient enrollment
-
Investigational product management
-
Data collection and monitoring
Experienced project managers oversee coordination between sponsors, CROs, sites, and regulatory bodies to ensure timelines remain intact.
Operational excellence during execution reduces enrollment delays and improves data quality—two of the most common challenges in clinical research.
4. Monitoring & Controlling: Ensuring Compliance and Quality
Monitoring runs in parallel with execution and focuses on maintaining compliance, tracking KPIs, and managing risks.
Modern approaches incorporate:
-
Risk-based monitoring (RBM)
-
Centralized data review
-
Quality management systems
-
Audit preparedness
-
Change control procedures
Regulatory documentation and reporting require precision. This is where expert medical writing services play a critical role—ensuring protocols, CSRs, and regulatory submissions meet global standards.
Learn more about CUREXBIO’s specialized Medical Writing Services for compliant, submission-ready documentation.
5. Closing: Regulatory Submission and Knowledge Transfer
The closing phase finalizes all deliverables, including:
-
Database lock
-
Clinical Study Report (CSR) preparation
-
Regulatory submission packages
-
Trial master file (TMF) archiving
-
Lessons learned documentation
Proper closure ensures smooth transition into post-marketing surveillance or subsequent trial phases.
Well-managed project closure strengthens long-term development strategy and accelerates future submissions.
How Integrated Clinical Project Management Reduces Risk
When lifecycle phases operate in silos, risks multiply. Integrated management ensures:
-
Early feasibility alignment
-
Transparent stakeholder communication
-
Real-time risk tracking
-
Budget control
-
Compliance consistency
CUREXBIO delivers end-to-end solutions across preclinical research, clinical development, and regulatory documentation, enabling sponsors to maintain control from molecule to market.
Visit CUREXBIO to explore comprehensive support solutions tailored to biotech, pharmaceutical, and research organizations.
Key Benefits of Partnering with an Experienced Clinical Development Team
-
Faster study startup timelines
-
Improved patient enrollment rates
-
Reduced protocol deviations
-
Enhanced regulatory readiness
-
Stronger data integrity
-
Streamlined global coordination
In an industry where delays can cost millions, strategic clinical project management is not optional—it’s essential.
Final Thoughts
From preclinical support services to clinical trial execution and regulatory submission management, a structured lifecycle approach ensures operational excellence at every stage.
Organizations that invest in strong clinical project management frameworks gain a competitive advantage—accelerating innovation while maintaining compliance and quality.
If you're looking to strengthen your development strategy, explore how CUREXBIO’s integrated solutions can support your next clinical milestone.
What's Your Reaction?
 Like
0
Like
0
 Dislike
0
Dislike
0
 Love
0
Love
0
 Funny
0
Funny
0
 Angry
0
Angry
0
 Sad
0
Sad
0
 Wow
0
Wow
0